 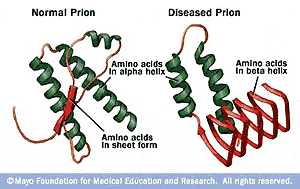
Under most circumstances, misfolded proteins are either refolded by chaperone proteins or degraded by the proteasome. Understanding these reversible amyloids can help us develop treatments for irreversible amyloid aggregates.Īmyloid fibrils are aggregates of misfolded proteins. Moreover, eukaryotes build reversible amyloid fibrils to pack and store secretory proteins until the cell needs to release them. Some bacteria use amyloid fibrils on their surfaces to create protective biofilms. However, not all amyloid fibrils are harmful. This transmissible form of amyloid formation is associated with fatal neurodegeneration. The misfolded PrPs contain beta-sheets, and thus tend to aggregate and form amyloid fibrils. All PrPs eventually assume the aberrant structure. Misfolded PrPs can convert normal PrPs into abnormal shapes. One particular prion protein, PrP, is a neural membrane protein. Prion diseases, such as Creutzfeldt-Jacob disease in humans and Bovine spongiform encephalopathy, commonly known as mad cow disease, also involve the formation of amyloid fibrils. These filaments can aggregate to form amyloid fibrils.Īccumulation of amyloid fibrils has been observed in certain neurodegenerative conditions, such as in Alzheimer's and Parkinson's diseases. In this structure, individual beta-sheets lie perpendicular to the central axis. Two closely-packed stacks of beta-sheets associate to form a cross-beta filament. Hundreds of beta-sheets in identical misfolded proteins form hydrogen bonds and stack to form long filaments. Some of these hydrophobic segments that normally fold into alpha-helices can assemble into beta-sheets. Whatever the mechanism, misfolding can expose short, hydrophobic segments of a protein causing it to become insoluble in water. Mutations can cause protein misfolding if the original protein shape becomes less favorable.Įxtrinsic factors, such as physical or chemical changes in the cytoplasm, force properly-folded proteins into new structures, suitable for the new environment. Inadequate cellular oversight, such as non-functioning chaperones or proteasomes due to aging or disease, can cause proteins to stay in abnormal shapes. Sometimes proteins fold into incorrect shapes, often referred to as misfolded proteins, which are degraded by the proteasome. Some proteins fold easily on their own but many are guided to fold correctly by proteins called chaperones. Proteins fold into energetically-favorable structures, with their hydrophobic amino acids on the inside and their charged and polar amino acids on the outside. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
